|
 Drug safety is in the interests of all
stakeholders: pharmaceutical companies, regulators, and the
patients who they serve.
Drug safety is in the interests of all
stakeholders: pharmaceutical companies, regulators, and the
patients who they serve.
Traditionally, statisticians have found the analysis of clinical
trial safety data to be difficult and plagued with problems.
At Data Clarity Consulting, we use modern statistical
attitudes and technologies to apply state
of the art data analysis to gain maximum insight from clinical
trial safety data.
Having identified methods commonly used in fields outside
of the pharmaceutical industry, and evaluated those methods
extensively on historical clinical trial data, we now have
a data analysis toolbox specifically designed for identifying the
potential for a new drug to cause toxic effects.
With the Mercury system for clinical
trial data review, we enable clinical reviewers to quickly and
easily gain insights into their data, avoiding the slow and
labour intensive reliance on summary tables and data listings.
With extreme value modelling, we
can characterize the frequency and magnitude of outliers in
laboratory and vital signs data starting with Phase I.
By establishing the strength of evidence for a treatment
impact on liver enzymes or other safety parameters, and
by using Bayesian simulation methods to predict future
toxicity events, we enable better informed decisions
and proper risk/benefit assessment.
With data mining, we can
systematically rank adverse events according to the strength
of evidence for a treatment difference, guiding clinical
reviewers to the unexpected events they need to investigate,
saving valuable time and making sure that no stone is left
unturned. We can then establish
whether or not those events appear to be related to age, body
weight, or other factors.
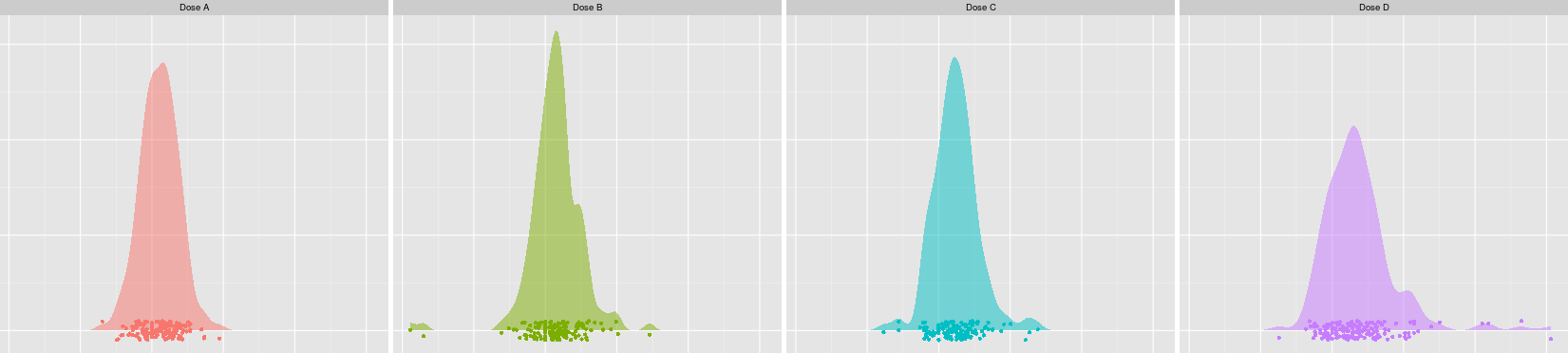
With appropriate
graphical displays,
we can enable clinical
and scientific colleagues to quickly see relationships between
treatments and safety parameters.
|